
A Lesson in Animal Model Choice: Uncovering the Role of Trem2 in Post-Stroke Neuroinflammation
In a study published in PNAS, Prof. ZHU Xinzhou's team from the Shenzhen Institutes of Advanced Technology of the Chinese Academy of Sciences, along with collaborators from Shenzhen University of Advanced Technology, revealed the mechanism of how the triggering receptor expressed on myeloid cells 2 (Trem2) exacerbates brain injury following ischemic stroke by amplifying microglia-mediated neuroinflammation.
The complexity of brain disorders makes it difficult for any single animal model to fully recapitulate disease pathology. Trem2 is a key immune receptor predominantly expressed on microglia. Genetic variants of Trem2 are strongly linked to Alzheimer's disease, where Trem2 promotes amyloid clearance and neuroprotection, with animal experiment results largely consistent with clinical data.
Although most animal studies suggest a neuroprotective role of Trem2, clinical evidence indicates that elevated soluble Trem2 in patient blood correlates with worse outcomes and higher mortality. This discrepancy highlights a critical gap between experimental models and human disease.
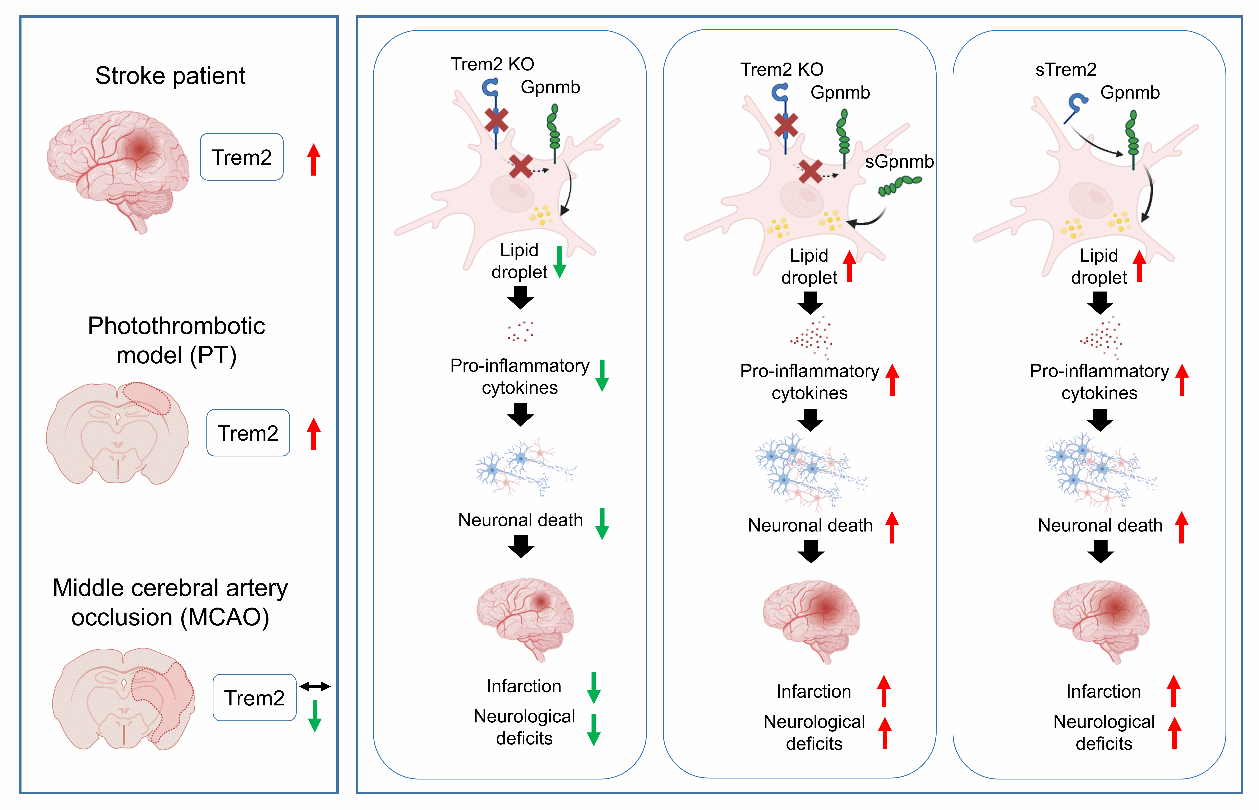
In this study, the researchers first re-evaluated the widely used middle cerebral artery occlusion (MCAO) model, and found that it failed to reproduce the marked upregulation of Trem2 observed in stroke patients. As a result, the model provided limited support for a protective role of Trem2.
Then, the researchers employed the photothrombotic model, and found that it induced robust neuroinflammation and showed strong Trem2 upregulation, closely matching clinical features. They also found that Trem2 deficiency reduced microglia-mediated inflammation, neuronal loss, and neurological deficits, while soluble Trem2 aggravated injury. These results were consistent with clinical observations.
Integrated transcriptomic analyses identified Gpnmb as a conserved downstream effector of Trem2. Functional validation confirmed that soluble Gpnmb counteracted the protective effects of Trem2 deficiency, while clinical data suggested that circulating Trem2 and Gpnmb might serve as the biomarkers of stroke severity.
The Trem2-Gpnmb axis drives neuroinflammation and worsens stroke outcomes. This study highlights that Trem2 may shift from protective in chronic disease to detrimental in acute injury, emphasizing the need for careful model selection to ensure translational relevance.
Graphic summary.(Image by SIAT)
File Download: